Background: Umbralisib is an oral, once-daily, dual inhibitor of phosphatidylinositol-3-kinase-delta (PI3Kδ) and casein kinase-1ε (CK1ε) that exhibits improved selectivity for the delta isoform of PI3K. The UNITY-NHL trial (NCT02793583) is a multicenter, open-label, registration directed Phase 2 study designed to evaluate the safety and efficacy of umbralisib in previously treated NHL patients (pts). Previously reported results in pts with relapsed/refractory (R/R) marginal zone lymphoma (MZL) demonstrated that umbralisib was active with a manageable safety profile. In contrast with other PI3K inhibitors, there was a low incidence of immune-mediated toxicities with umbralisib, possibly attributable to enhanced selectivity for the PI3Kδ isoform as well as inhibition of CK1ε. Herein, we present results from the final analysis of the iNHL population treated with single agent umbralisib.
Methods: Eligible pts had histologically confirmed iNHL: MZL (splenic, nodal, extranodal), follicular lymphoma (FL; Gr 1, 2, 3a), or small lymphocytic lymphoma (SLL). MZL pts were R/R to ≥1 prior lines of treatment, which must have included an anti-CD20, while FL and SLL pts were R/R to ≥2 prior lines, which had to include an anti-CD20 and an alkylating agent. Umbralisib was administered orally at 800 mg once-daily in 28-day treatment cycles until disease progression or unacceptable tolerability. The primary endpoint of the study was overall response rate (ORR) as assessed by an independent review committee (IRC), according to the revised IWG criteria (Cheson 2007). Secondary endpoints included duration of response (DoR), progression-free survival (PFS), time to response (TTR), and safety. Pneumocystis jiroveci pneumonia (PCP) and anti-viral prophylaxis were mandated for all pts.
Results: 208 iNHL pts received at least 1 dose of umbralisib, including 69 MZL, 117 FL, and 22 SLL pts. The median duration of exposure was 8.4 mos (range 0.2 - 27.0), median age was 66, and 56.7% were male. Pts had received a median of 2 prior regimens (range 1 - 10) with 46.1% having received ≥ 3 regimens. FL patients had a median of 3 prior regimens.
With a median follow up of 27.8 mos, MZL pts had an ORR of 49.3% (95% CI 37.0% - 61.6%) with 15.9% achieving a complete response (CR), and a Disease Control Rate (DCR: CR+PR+SD) of 82.6%. ORR was consistent amongst MZL subtypes. The median TTR was 2.8 months (95% CI 2.7 - 2.9). The median profession-free survival (PFS) was not reached (95% CI 12.1 mos - not evaluable [NE]) with an estimated 12-month PFS rate of 64.2%. The median DoR was not reached (95% CI 10.3 mos - NE), and no pts who achieved CR have experienced disease progression to date.
With a median follow up of 27.5 mos, FL pts had an ORR of 45.3% (95% CI 36.1% - 54.8%) with 5.1% achieving a CR, and a DCR of 79.5%. The median TTR was 4.6 mos (95% CI 3.0 - 5.6). The median PFS was 10.6 mos (95% CI 7.2 - 13.7) with an estimated 12-month PFS rate of 45.9%. The median DoR was 11.1 mos (95% CI 8.3 - 15.6).
With a median follow up of 29.3 mos, SLL pts had an ORR of 50.0% (95% CI 28.2 - 71.8) with 4.5% achieving a CR, and a DCR of 86.4%. The median TTR was 2.7 mos (95% CI 2.4 - 2.8). The median PFS was 20.9 mos (95% CI 7.4 - 24.1) with an estimated 12-month PFS rate of 62.6%. The median DoR was 18.3 mos (95% CI 2.4 - NE).
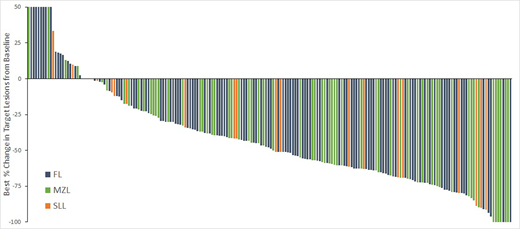
Best % change in target lesions form baseline for pts with at least one post-baseline assessment is shown in the figure.
At the data cut-off, 60 pts (26 MZL, 27 FL, 7 SLL) remained on treatment. The most common ≥G3 AEs were neutropenia (11.5%), diarrhea (10.1%) and increased ALT/AST (7.2%). Other AEs of interest included pneumonitis (All G 1.4%; ≥G3 1.0%), and colitis (All G 1.4%; ≥G3 0.5%). Serious AEs were reported in 28.1% of pts, with 24.6% ≥G3. One patient with SLL had a fatal myocardial infarction (unrelated to umbralisib); there were no other G5 AEs. A total of 31 pts (14.9%) discontinued due a treatment-related adverse event (AE). Treatment-related AEs leading to dose reductions occurred in 20 (9.6%) pts.
Conclusions: In the Phase 2 UNITY-NHL study, umbralisib achieved meaningful clinical activity in a heavily pretreated iNHL population. The safety profile was manageable, with a relatively low incidence of immune-mediated toxicities and AE-related discontinuations. These results suggest umbralisib has a favorable benefit-risk profile in this patient population and further development is ongoing.
Zinzani:Kirin Kyowa: Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Merck: Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; BMS: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Kyowa Kirin: Consultancy, Speakers Bureau; MSD: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; ADC Therapeutics: Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Celgene: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Immune Design: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Portola: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Eusapharma: Consultancy, Speakers Bureau; Incyte: Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Takeda: Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Gilead: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; TG Therapeutics, Inc.: Honoraria, Speakers Bureau; Servier: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Immune Design: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Verastem: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Janssen-Cilag: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; EUSA Pharma: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Sanofi: Consultancy, Membership on an entity's Board of Directors or advisory committees; AbbVie: Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Janssen: Consultancy, Honoraria, Speakers Bureau; Roche: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Sandoz: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Celltrion: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau. Jurczak:Sandoz-Novartis: Consultancy; Afimed: Research Funding; Acerta: Consultancy, Research Funding; Pharmacyclics: Research Funding; Bayer: Research Funding; Maria Sklodowska-Curie National Research Institute of Oncology, Krakow, Poland: Current Employment; TG Therapeutics, Inc.: Research Funding; Celgene: Research Funding; Servier: Research Funding; Takeda: Research Funding; Roche: Research Funding; Merck: Research Funding; MEI Pharma: Research Funding; Janssen China R&D: Consultancy, Research Funding; BeiGene: Consultancy, Research Funding; AstraZeneca: Consultancy; European Medicines Agency,: Consultancy; Jagiellonian University, Krakow, Poland: Ended employment in the past 24 months; Epizyme: Consultancy; Gilead Sciences: Research Funding; MorphoSys: Research Funding; Nordic Nanovector: Research Funding. Ghosh:Kite/Gilead: Consultancy, Speakers Bureau; Juno/Celgene/Bristol-Myers Squibb: Consultancy, Research Funding; Pharmacyclics LLC, an AbbVie Company: Consultancy, Research Funding, Speakers Bureau; SGN: Consultancy, Research Funding, Speakers Bureau; TG Therapeutics: Consultancy, Research Funding; Roche/Genentech: Research Funding; Karyopharm: Consultancy; Genmab: Consultancy, Speakers Bureau; AbbVie: Speakers Bureau; AstraZeneca: Speakers Bureau; Celgene/Bristol-Myers Squibb: Speakers Bureau; Forty Seven Inc: Consultancy, Other: Research Bureau, Research Funding; Janssen: Consultancy, Research Funding, Speakers Bureau. Derenzini:TG Therapeutics, Inc.: Research Funding. Cheah:Celgene, F. Hoffmann-La Roche, MSD, Janssen, Gilead, Ascentage Pharma, Acerta, Loxo Oncology, TG therapeutics: Honoraria; Celgene, F. Hoffmann-La Roche, Abbvie, MSD: Research Funding. Phillips:Karyopharm: Consultancy; Beigene: Consultancy; Seattle Genetics: Consultancy; BMS: Consultancy; Abbvie: Consultancy, Research Funding; AstraZeneca: Consultancy; Incyte: Consultancy, Other: travel expenses; Cardinal Health: Consultancy; Bayer: Consultancy, Research Funding; Pharmacyclics: Consultancy. Lech-Marańda:Roche, Amgen, Gilead: Speakers Bureau; Roche, Novartis, Takeda, Janssen-Cilag, Amgen, Gilead, AbbVie, Sanofi: Consultancy. Cheson:Abbvie: Consultancy, Research Funding; Kite: Consultancy; TG Therapeutics: Speakers Bureau; Morphosys: Consultancy; Symbio: Membership on an entity's Board of Directors or advisory committees; Karyopharm: Consultancy, Membership on an entity's Board of Directors or advisory committees; Pharmacyclics: Consultancy, Research Funding; Jannsen: Consultancy; Parexel: Consultancy; Trillium: Research Funding; GSK: Membership on an entity's Board of Directors or advisory committees. Caimi:Verastem: Other: Advisory Board; Celgene: Speakers Bureau; Amgen: Other: Advisory Board; Bayer: Other: Advisory Board; Kite Pharma: Other: Advisory Board; ADC Therapeutics: Other: Advisory Board, Research Funding. Leslie:BeiGene: Honoraria, Speakers Bureau; KitePharma: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Seattle Genetics: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Pharmacyclics: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; BMS: Speakers Bureau; Celgene: Speakers Bureau; Janssen: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; TG Therapeutics: Consultancy, Membership on an entity's Board of Directors or advisory committees; AbbVie: Consultancy, Membership on an entity's Board of Directors or advisory committees; ADC therapeutics: Consultancy, Membership on an entity's Board of Directors or advisory committees; Bayer: Consultancy, Membership on an entity's Board of Directors or advisory committees; Karyopharm: Honoraria, Speakers Bureau; Epizyme: Honoraria, Speakers Bureau; AstraZeneca: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau. Chavez:Karyopharm: Consultancy; BeiGene: Speakers Bureau; Bayer: Consultancy; Merck: Research Funding; AbbVie: Consultancy; Genentech: Speakers Bureau; Epizyme: Speakers Bureau; Gilead: Consultancy; Novartis: Consultancy; Kite, a Gilead Company: Consultancy, Speakers Bureau; Celgene: Consultancy; Morphosys: Consultancy, Speakers Bureau; AstraZeneca: Speakers Bureau; Verastem: Consultancy; Pfizer: Consultancy. Fonseca:Bristol-Myers Squibb Company: Speakers Bureau. Babu:Merck: Research Funding; Syndax: Research Funding; AbbVie: Research Funding; Janssen Oncology: Research Funding; TG Therapeutics: Research Funding; Amgen: Research Funding; Sanofi: Research Funding; Lutheran Hospital: Other; Argenx: Consultancy, Research Funding; Nektar: Research Funding; Novartis: Research Funding; Bayer: Honoraria; Lilly: Honoraria, Other: TRAVEL, ACCOMMODATIONS, EXPENSES (paid by any for-profit health care company), Research Funding; Alexion Pharmaceuticals: Consultancy, Honoraria, Other: TRAVEL, ACCOMMODATIONS, EXPENSES (paid by any for-profit health care company), Research Funding, Speakers Bureau; Bristol-Myers Squibb: Consultancy, Honoraria, Other: TRAVEL, ACCOMMODATIONS, EXPENSES (paid by any for-profit health care company), Research Funding; Fort Wayne Medical Oncology & Hematology: Current Employment, Current equity holder in publicly-traded company; AstraZeneca/MedImmune: Research Funding; AstraZeneca: Consultancy, Honoraria; Genentech, Inc./ F. Hoffmann-La Roche: Research Funding; Boehringer Ingelheim: Consultancy. Hodson:Gilead Sciences: Research Funding. Burke:Bristol Myers Squibb: Consultancy; Verastem: Consultancy; Bayer: Consultancy; Astra Zeneca: Consultancy; Gilead: Consultancy; Seattle Genetics: Speakers Bureau; AbbVie: Consultancy; Morphosys: Consultancy; Adaptive: Consultancy; Epizyme: Consultancy; Kura: Consultancy; Celgene: Consultancy; Adaptive Biotechnologies: Consultancy; Roche: Consultancy. Sharman:TG Therapeutics: Consultancy, Research Funding; Bristol Meyers Squibb: Consultancy, Research Funding; BeiGene: Research Funding; Genentech: Consultancy, Research Funding; AbbVie: Consultancy, Research Funding; Pfizer: Consultancy, Research Funding; Pharmacyclics: Consultancy, Research Funding; AstraZeneca: Consultancy, Research Funding; Acerta: Consultancy, Research Funding; Roche: Consultancy, Research Funding; Celgene: Consultancy, Research Funding. O'Connor:Kymera Therapeutics: Current equity holder in private company, Honoraria, Membership on an entity's Board of Directors or advisory committees; Celgene: Honoraria, Other: Data Safety Monitoring Committee, Research Funding; TG Therapeutics: Current Employment, Current equity holder in publicly-traded company; Servier: Consultancy; Mundipharma: Other: Consulting; Nomocan: Membership on an entity's Board of Directors or advisory committees; Merck: Research Funding; Astex Pharmaceuticals: Honoraria, Research Funding. Miskin:TG Therapeutics, Inc.: Current Employment, Current equity holder in publicly-traded company. Sportelli:TG Therapeutics, Inc.: Current Employment, Current equity holder in publicly-traded company. Weiss:TG Therapeutics, Inc.: Current Employment, Current equity holder in publicly-traded company, Membership on an entity's Board of Directors or advisory committees. Fowler:Celgene: Membership on an entity's Board of Directors or advisory committees, Research Funding; Roche: Research Funding; TG Therapeutics: Membership on an entity's Board of Directors or advisory committees, Research Funding; Novartis: Membership on an entity's Board of Directors or advisory committees, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.